Clinical Value of Quantative Pupillometry in Stroke Care
Exploring how quantitative pupillometry enhances stroke monitoring and improves early detection of neurological decline.
Exploring how quantitative pupillometry enhances stroke monitoring and improves early detection of neurological decline.

.png)
Stroke persists as a formidable public health crisis in the United States, imposing a substantial burden through high rates of mortality and significant, often devastating, long-term disability. In 2022 alone, stroke accounted for 1 in every 6 deaths attributable to cardiovascular disease nationwide. The sheer frequency is alarming: every 40 seconds, someone in the US experiences a stroke, and every 3 minutes and 11 seconds, someone succumbs to one. The age-standardized prevalence of self-reported stroke further underscores this escalating challenge, having risen to 2.9% in the 2020–2022 period. This epidemiological landscape translates into more than 795,000 individuals having a stroke each year, with approximately 610,000 of these being first-time events and a concerning nearly 1 in 4 (approximately 185,000) occurring in individuals who have had a previous stroke. Ischemic strokes, resulting from blocked blood flow to the brain, constitute the vast majority, accounting for about 87% of all stroke cases.
Stroke encompasses both ischemic events, which are caused by the occlusion of cerebral blood vessels, and hemorrhagic events, which result from the rupture of these vessels. Irrespective of the primary vascular insult, both pathways can trigger a cascade of secondary brain injury mechanisms that are critical determinants of the patient's ultimate neurological recovery and functional capacity. The dynamic and often unpredictable nature of these secondary injuries necessitates continuous and vigilant neuromonitoring. The primary objective of such intensive monitoring is the proactive identification and effective mitigation of factors that have the potential to exacerbate the initial brain damage, principally through the meticulous optimization of cerebral perfusion and metabolic function. While traditional methods of clinical neurological assessment, including widely utilized instruments such as the Glasgow Coma Scale (GCS), form an indispensable foundation of neurological evaluation, their practical utility can be significantly constrained in certain challenging clinical scenarios. These include situations where patients are under the influence of deep sedation or when they present with profound neurological impairments from the very onset of their illness. A notable and clinically relevant limitation of the GCS, particularly during the hyperacute and prehospital phases of stroke management, is its inherent omission of specific assessments for critical brainstem reflexes, most importantly, the detailed evaluation of pupillary light responses (Robba et al., 2024).
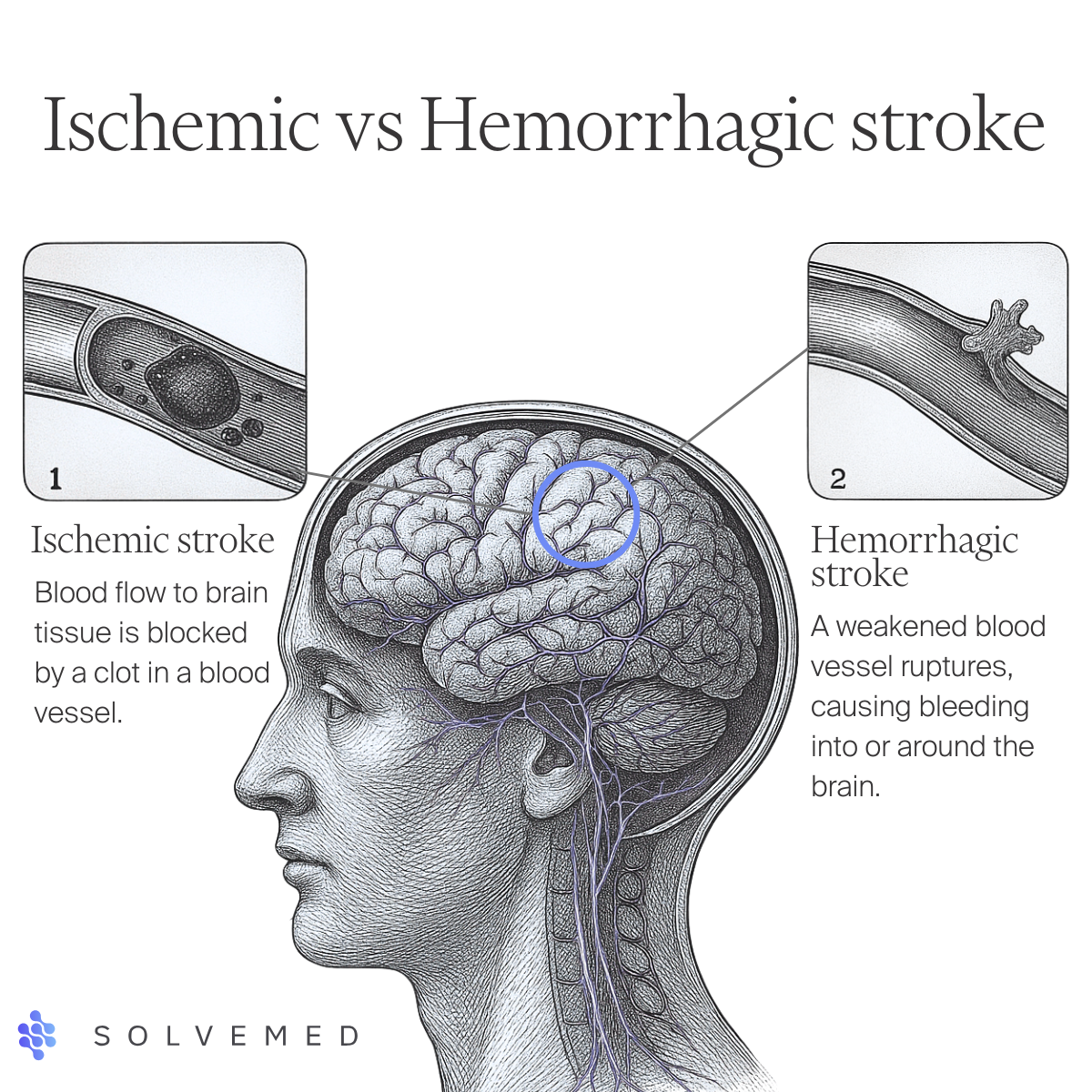
Stroke is broadly classified into ischemic phenomena, resulting from the occlusion of cerebral vasculature, and hemorrhagic events, characterized by the rupture of a blood vessel within the cranial vault. Although these primary insults differ in their vascular origin, both ischemic and hemorrhagic strokes initiate common pathways of secondary brain injury, which are major determinants of long term neurological recovery and functional independence (Robba et al., 2024). The evolving and often unpredictable nature of these secondary injury cascades underscores the need for continuous, vigilant neuromonitoring. The overarching aim of such monitoring is to proactively identify and counteract factors that may exacerbate the initial brain damage, primarily through the meticulous optimization of cerebral perfusion and metabolic homeostasis (Robba et al., 2024). Traditional methods of clinical neurological assessment, including widely used instruments like the Glasgow Coma Scale (GCS), form the bedrock of neurological evaluation. However, their practical utility can be limited in specific clinical scenarios, such as when patients are under deep sedation or present with profound neurological impairments from the onset of their illness (Robba et al., 2024). A pertinent limitation of the GCS, particularly in the hyperacute and prehospital phases of stroke care, is its exclusion of specific assessments for critical brainstem reflexes, most notably pupillary light responses (Robba et al., 2024).
Quantitative pupillometry has emerged as a significant technological advancement, offering an objective, standardized, and highly reproducible method for the comprehensive evaluation of the pupillary light reflex (PLR). This innovation effectively addresses and overcomes the inherent subjectivity and potential for inaccuracies that can often compromise traditional manual penlight examinations of the pupils (Robba et al., 2024; McNett et al., 2017). Contemporary quantitative pupillometers are meticulously engineered to precisely measure a wide spectrum of PLR characteristics. These include critical static parameters such as the baseline pupil size, as well as dynamic parameters like the velocity of pupillary constriction (CV) in direct response to a carefully calibrated light stimulus. The rich, objective data points generated by these devices are frequently integrated by sophisticated proprietary algorithms into a single composite score. While this score is often referred to in the broader scientific literature as the Pupil Light Reflex (PLR), for the purpose of maintaining clarity and consistency within this clinical discussion, it will be termed the Pupillary Light Reflex (PLR) value (McNett et al., 2017; Aoun et al., 2019; Privitera et al., 2022). In accordance with the current body of scientific literature and prevailing manufacturer guidelines, a PLR value that falls within the numerical range of 3 to 5 is generally interpreted as being within normal physiological limits. Conversely, PLR values that register below the threshold of 2.9 may serve as an indicator of the presence of an underlying pathological process that is adversely affecting the integrity of the pupillary reflex pathways or, more broadly, overall neurological function (Aoun et al., 2019; Privitera et al., 2022).
An extensive and growing body of scientific investigation has clearly illuminated several pivotal and highly relevant clinical applications of quantitative pupillometry within the framework of comprehensive stroke management. One of its most clinically impactful and widely recognized contributions is its demonstrated and reliable capacity for the early detection of incipient, or gradually developing, neurological deterioration. Multiple rigorous research studies have consistently and compellingly reported that subtle, yet quantitatively discernible, alterations in various PLR parameters, as meticulously captured and analyzed by quantitative pupillometry, can often precede the overt clinical manifestation of developing ischemic brain injury or impending brain herniation syndromes by a clinically significant margin. This lead time can frequently extend to several hours, providing a crucial window for intervention (Aoun et al., 2019; Osman et al., 2019). This prodromal period, which is identified through objective pupillometric changes, offers clinicians a critical and highly actionable window of opportunity. During this valuable interval, preemptive diagnostic evaluations and targeted therapeutic interventions can be initiated with the primary aim of mitigating the potential severity, or even averting the ultimate progression, of irreversible neurological sequelae (Aoun et al., 2019). For example, in the specific and often particularly challenging context of managing patients who have suffered an aneurysmal subarachnoid hemorrhage, a severe and often devastating subtype of hemorrhagic stroke, detailed investigations have documented that a discernible decline in PLR values to ranges considered abnormal was observable more than eight hours prior to any clinically apparent neurological decline in 71.4% of the studied cases where such a subsequent clinical decline eventually occurred (Aoun et al., 2019).
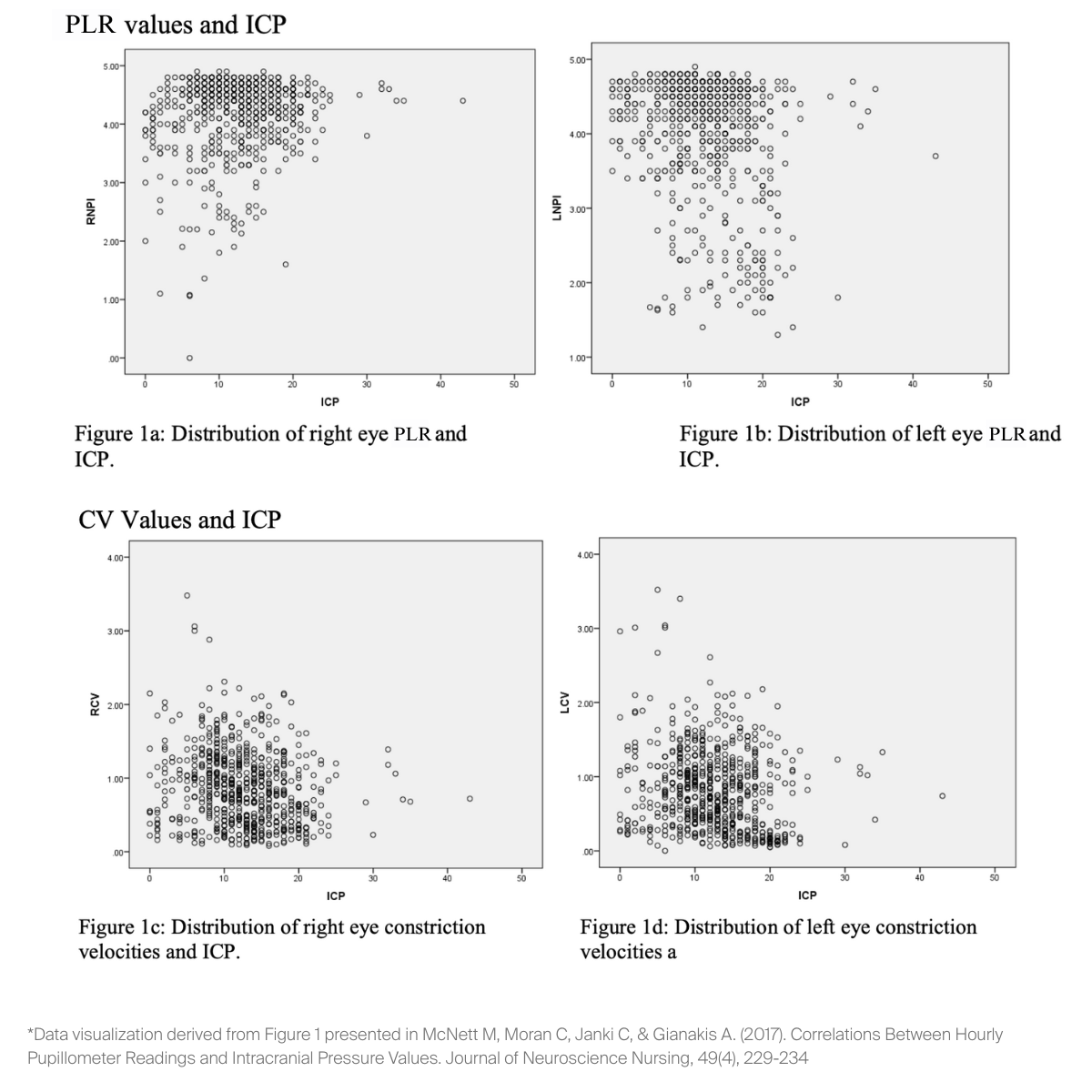
Furthermore, the quantitative data derived from quantitative pupillometry have demonstrated robust and clinically meaningful correlations with several critical physiological variables that accurately reflect the dynamics of intracranial pathophysiology. These variables most notably include intracranial pressure (ICP) and the extent of cerebral midline shift. Thorough investigations that have been conducted across diverse and representative populations of neurocritically ill patients have firmly established a statistically significant and clinically relevant relationship between various pupillometer derived metrics, such as the calculated PLR values and the measured pupillary constriction velocity, and concurrently monitored ICP levels (McNett et al., 2017). One notable prospective cohort study involving 76 neurocritically ill subjects found that hourly pupillometer readings, including constriction velocity and PLR values, significantly correlated with ICP values in both bivariate analyses (with correlation coefficients ranging from 0.13 to 0.23, and all p values < .001) and in more complex multivariate regression models (F(6)=17.63, p < .001), highlighting the adjunctive value of pupillometry to traditional invasive monitoring (McNett et al., 2017). In a parallel and complementary line of scientific inquiry, objective pupillary data, with particular emphasis placed on the PLR value and the dynamic pupillary constriction velocity, have been clearly shown to correspond significantly with the measured magnitude of horizontal intracranial midline displacement. This structural shift is a critical indicator of intracranial mass effect and is commonly observed in patients suffering from both ischemic and hemorrhagic stroke subtypes (Osman et al., 2019). Specifically, a study by Osman et al. (2019) involving 134 stroke patients demonstrated that after controlling for potential confounding variables such as age, race, and gender, there was a significant correlation between the shift of the septum pellucidum (SPS), a key marker of midline shift, and the PLR (for the left pupil, p < .001; for the right pupil, p < .001), as well as with the constriction velocity (for the left pupil, p < .005; for the right pupil, p < .001). The early and accurate detection of an evolving or progressive midline shift is of paramount clinical importance, as it frequently signifies an escalating intracranial mass effect, which, if not promptly and effectively addressed, can herald further devastating secondary brain injury and contribute to poorer patient outcomes.
In the specialized and often complex management of patients who have experienced an aneurysmal subarachnoid hemorrhage, quantitative pupillometry provides invaluable adjunctive diagnostic information, particularly for the critical task of monitoring for the potential development of delayed cerebral ischemia (DCI). DCI remains a common and feared complication in this specific patient population, and it contributes significantly to both increased morbidity and mortality rates. While transcranial Doppler (TCD) ultrasonography is routinely and widely employed as a non invasive screening tool for the detection of cerebral vasospasm, which is recognized as a frequent precursor or risk factor for DCI, the predictive accuracy of TCD in identifying precisely which patients will ultimately progress to develop clinically significant DCI is acknowledged to be somewhat limited (Aoun et al., 2019). There is an emerging and compelling body of evidence which indicates that specific pupillometric changes, most notably a quantifiable and sustained reduction in the PLR value, have demonstrated a strong and statistically significant association with the eventual development of DCI (Aoun et al., 2019). This important finding suggests that the incorporation of serial quantitative pupillometry into the monitoring protocol could potentially serve as an earlier and perhaps more specific marker of impending clinical deterioration that is directly related to DCI in this highly vulnerable cohort of patients (Aoun et al., 2019). In a study focused on this patient group, Aoun et al. (2019) reported a significant association between the occurrence of DCI and an abnormal decrease in the PLR value, with an odds ratio of 3.3930 (95% CI 2.2789–5.0517, p < 0.001), indicating a strong predictive link. It is particularly noteworthy from a clinical perspective that the presence of isolated sonographic evidence of vasospasm, as detected by TCD, does not invariably or consistently correlate with concurrent changes in PLR parameters. This observed dissociation between vascular imaging findings and pupillary function lends substantial support to the hypothesis that alterations in pupillary responsiveness, as accurately captured and quantified by quantitative pupillometry, are more directly and reliably indicative of actual ischemic neuronal injury occurring at the tissue level, rather than merely reflecting the presence of narrowed or spastic cerebral blood vessels (Aoun et al., 2019).
The prognostic utility of quantitative pupillometry in the diverse population of stroke patients represents another important dimension of its overall clinical value, and this area is currently the subject of active and ongoing research and clinical refinement. The specific concept of a Pupillary Light Reflex (PLR) differential, which is quantitatively defined as a significant and measurable disparity (typically characterized by an absolute difference of ≥ 0.7) between the PLR values that are recorded from the left and the right pupils, has been prospectively investigated. These investigations have linked the presence of such a differential with less favorable functional outcomes at the time of hospital discharge, as objectively and systematically measured by the modified Rankin Scale (mRS), in broad cohorts of patients who have experienced various types of stroke (Privitera et al., 2022). For example, a large registry study demonstrated that stroke patients who exhibited at least one occurrence of a significant PLR differential (defined as a difference ≥ 0.7 between the eyes) during their stay in the neuro-intensive care unit had notably higher, indicating worse, discharge mRS scores (with a mean mRS of 3.9) when compared to similar stroke patients who did not exhibit such a PLR differential (who had a mean mRS of 2.7; P < .001) (Privitera et al., 2022). Furthermore, a Receiver Operating Characteristic (ROC) curve analysis conducted to determine the optimal cutoff point for this PLR differential yielded an Area Under the Curve (AUC) of 0.71 (P < .001), statistically confirming that a difference of ≥ 0.7 is a clinically relevant and informative threshold for identifying patients at higher risk of poor outcome (Privitera et al., 2022). This observed association between significant pupillary asymmetry and poorer functional outcomes appears to hold true and maintain its significance even in those clinical situations where the individual PLR scores for each eye, when they are considered in isolation from each other, remain within the accepted normal physiological range. This critical finding strongly suggests that the presence of a quantifiable asymmetry in pupillary responsiveness, as can be precisely detected and reliably quantified only by quantitative pupillometry, may itself serve as an independent and clinically significant prognostic indicator. This can provide additional and valuable information for more accurate risk stratification, individualized outcome prediction, and potentially for guiding therapeutic decision making in patients who have suffered a stroke (Privitera et al., 2022).
Beyond its direct and increasingly recognized diagnostic applications and its emerging and promising prognostic capabilities, quantitative pupillometry also makes a very significant and practical contribution by substantially enhancing the overall reliability and inherent objectivity of the serial neurological assessments that are routinely conducted at the patient's bedside. By consistently delivering precise, quantitative, and highly reproducible measurements of various dynamic aspects of pupillary function over time, this advanced technology effectively minimizes, if not entirely eliminates, the well documented issue of inter observer variability. This variability is an inherent and widely acknowledged limitation of traditional, subjective, manual pupillary examinations that are performed with a conventional penlight (Robba et al., 2024; McNett et al., 2017; Osman et al., 2019). This fundamental characteristic of providing inherently objective and therefore more reliable data firmly positions quantitative pupillometry as an invaluable and increasingly indispensable component of any comprehensive, multimodal neuromonitoring strategy. Such sophisticated monitoring strategies are particularly essential within the specialized, dynamic, and high acuity environment of the modern neurocritical care unit, where the early detection of subtle neurological changes can have profound and immediate implications for patient management and ultimate clinical outcomes (McNett et al., 2017).
In synthesizing the current scientific landscape and the accumulating body of clinical evidence, it is unequivocally clear that quantitative pupillometry represents a significant and impactful technological advancement. This technology is progressively carving out an expanding and undeniably crucial role in the sophisticated, data driven, and increasingly personalized management of patients who are suffering from the myriad consequences of acute stroke. Its well documented and repeatedly demonstrated ability to furnish objective, early, and highly reliable indicators of an evolving neurological status and of impending clinical deterioration, coupled with its established and statistically significant correlations with critical pathophysiological parameters such as intracranial pressure and cerebral midline shift, and its emerging and promising prognostic capabilities related to long term functional outcome, collectively and strongly support its thoughtful and systematic integration into the standard of care for all eligible stroke patients. For clinicians who are at the forefront of stroke management, and who are continually striving to optimize therapeutic interventions, to personalize care pathways based on individual patient risk profiles, and ultimately to improve the long term neurological and functional outcomes for individuals who have been profoundly impacted by the complex and dynamic processes of acute stroke, quantitative pupillometry offers an increasingly indispensable, powerful, and clinically actionable tool.
Robba C, Zanier ER, Lopez Soto C, Park S, Sonneville R, Helbok R, et al.
Mastering the brain in critical conditions: an update. Intensive Care Med Exp. 2024;12(1):1–11.
doi:10.1186/s40635-023-00564-2
Privitera CM, Neerukonda SV, Aiyagari V, Yokobori S, Puccio AM, Schneider NJ, et al; END PANIC Investigators.
A differential of the left eye and right eye neurological pupil index is associated with discharge modified Rankin scores in neurologically injured patients. BMC Neurol. 2022;22:273.
doi:10.1186/s12883-022-02836-0
Osman M, Stutzman SE, Atem F, Olson D, Hicks AD, Ortega-Perez S, et al.
Correlation of objective pupillometry to midline shift in acute stroke patients. J Stroke Cerebrovasc Dis. 2019;28(7):1902–10.
doi:10.1016/j.jstrokecerebrovasdis.2019.03.018
McNett M, Moran C, Janki C, Gianakis A.
Correlations between hourly pupillometer readings and intracranial pressure values. J Neurosci Nurs. 2017;49(4):229–34.
doi:10.1097/JNN.0000000000000305
Aoun SG, Stutzman SE, Vo PN, El Ahmadieh TY, Osman M, Neeley O, et al.
Detection of delayed cerebral ischemia using objective pupillometry in patients with aneurysmal subarachnoid hemorrhage. J Neurosurg. 2019;1–6.